
Chemistry |
| Hydrolysis of Procyanidins |
Historically, winemakers
thought the distribution of various sized, condensed tannins extracted from
seeds and skins depended to a high degree on maturity of the grapes. However,
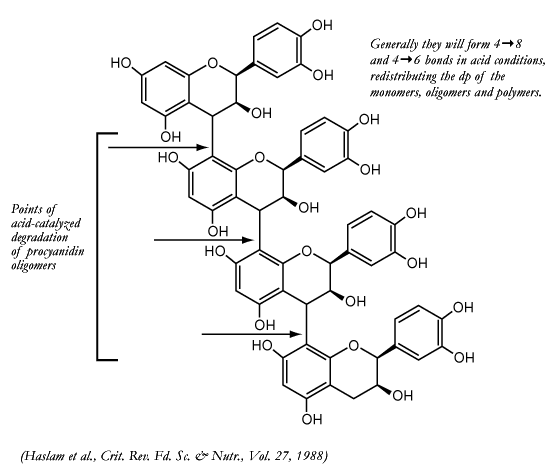
Haslam first suggested in 1980 and later wrote in a 1988 review paper, that
condensed tannins (polyphenolics) are not stable to hydrolysis. The conditions
found in wine (low pH, high acidic conditions) favor hydrolysis at the
4–>8 positions or the 4–>6 positions. Why is the
phenomena important?
|
Proposed mechanisms leading to T-A and T-T adducts
under
non-oxidative conditions.
Hydrolysis of tannin oligomers
or polymers is the central point a mechanism proposed by Véronique
Cheynier under reductive conditions. This mechanism helps to explain compounds
isolated from red wines and how these compounds may be formed. As Haslam has
suggested, a tannin molecule, e.g. dp 8 (8 catechin or epicatechin units) in
length, may be hydrolyzed under acidic conditions in wine. For example, as
shown in the figure below, this hydrolysis may produce two shorter oligomers,
dp 4 in length. However, the hydrolysis produces one neutral oligomer and one
positively charge oligomer (carbocation).
Depending on the
concentrations of available tannins or anthocyanins, the carbocation formed
will react with one or the other., If the reaction is with another tannin a
longer oligomer or polymer will be formed.
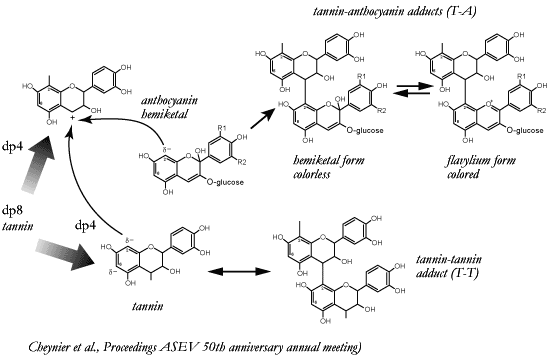
However, the process
differs if an anthocyanin is involved. First, note that the anthocyanin must be
in the hydrated or colorless form. This form provides an electron-rich molecule
which more readily reacts with the formed carbocation.
The
reaction occurs between the two molecules at the 4 and 8 positions and a
covalent bond is formed. Once formed, the tannin part acts as an electron sink
and favors the loss of water (the hydration of the anthocyanin) and a
stabilized color or anthocyanin-tannin adduct is formed.
Losing
the water of hydration has another unique aspect: the terminal molecule (the
anthocyanin) no longer has an excess of available electrons. Thus, the
anthocyanin acts as a terminus or terminal quencher for any further reaction at
this end of the oligomers or polymers being formed. |
| Proposed condensation reactions under non-oxidative
conditions |
|
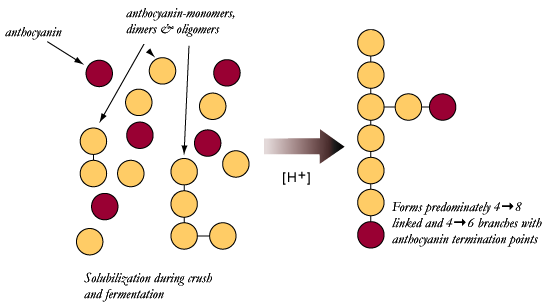
Here is a
more simplistic representation of the previous section. Reactions under
reductive conditions occur more slowly, depending on conditions and
availability of reactants.
However, scientists think that
tannins formed under these conditions will be longer, with a more uniform
structure. The longer and more uniform the tannins, the more likely there will
be a strong interaction between the tannin molecules.
This
interaction causes an association and aggregation of similar molecules
attempting to protect hydrophobic centers (the aromatic rings). Hydroxyls
located on the outer portions of the polymers will hydrogen bond, aiding in the
stabilization of the molecule, but at the same time excluding water
molecules.
Once the aggregate of molecules becomes large enough
and excludes enough water it will lose its ability to stay soluble and
precipitate from solution.
We see examples of this during
extended maceration, especially under highly reductive conditions. At first, it
appears that we are extracting more tannin and color. Yet within one to three
months, most of this material precipitates from the wine and color is
lost. |
| Summary of reactions under non-oxidative
conditions |
1. Under reductive conditions (no crosslinking by
acetaldehyde). A distribution of procyanidin oligomers and polymer sizes will
tend to redistribute to a median size. For example, range of sizes of dp 2 to
15, will tend to favor formation of a median size such as dp 7.
2. Anthocyanins will interfere with this process by
acting as terminal quenchers. Once an anthocyanin has linked to a terminal end
of a polymer or oligomer no further reactions with that molecule will occur,
other than losing the sugar moiety.
In model wine solutions, with
no interfering compounds, a distribution of procyanidin oligomers should, over
time, form an average size molecule.
However, if interfering
compounds such as anthocyanins are present, then the distribution of molecule
sizes will be much more unpredictable in manner. |
|
|
|







