
Chemistry |
Proposed role of
co-pigmentation
1. Anthocyanins contain aromatic rings
(hydrophobic) which are highly hydroxylated (hydrophillic).
2. The sugar esterified to the 3 position of the C ring
helps to maintain solubility
3. Must
conditions favor water soluble compounds; wine conditions are more favorable
for hydrophobic compounds.
4. Other
compounds present with similar properties will tend to aggregate and stack to
protect the hydrophobic centers.
5.
Conditions during later fermentation (increasing alcohol and temperature) favor
disruption of these aggregated or stacked complexes.
6. Presence of crosslinkers and close proximity of
anthocyanins and phenolics polymers (co-pigmentation) favors the crosslinking
reaction.
7. Toasted oak provides
compounds able to both co-pigment (stack) and crosslink, favoring the formation
of stabilized pigments.
|
|
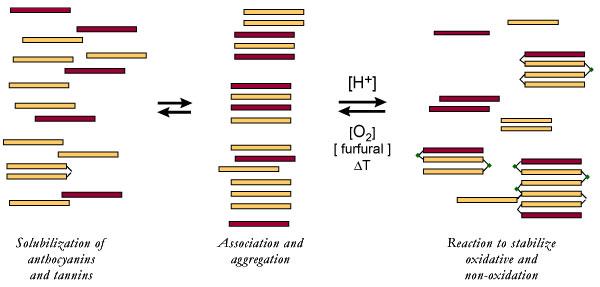
A pictorial of tannins and
anthocyanins solubilizing is on the left; stabilizing themselves through
stacking and aggregation is in the middle. On the right, (depending on
conditions), tannins and anthocyanins will either bond directly or crosslink if
acetaldehyde, furfural, HMF or 5-methyl furfural are present.
It
is the relative proximity and orientation of the molecules that may be the most
important role of co-pigmentation. This closeness and arrangement of the
molecules may aid in the crosslinking reaction, much in the manner that enzymes
work when catalyzing reactions between molecules.
|
|
|
|







